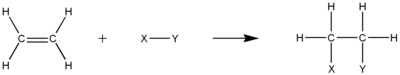why are the Acidity increases going down a row of the periodic table?
answer : according my opinion and acidity increases going left to right (because more electronegative atoms can stabilize the negative charge on the anion) and increases going down because the larger the atom the better it can delocalize the negative charge.
Nucleophicity and basicitiy tend to be similar to each other but opposite to electrongegativity trends. Nucleophicity increases going left across a period and up a group.
The Electron affinity of a molecule or atom is the energy change when an electron is added to the neutral atom to form a negative ion. This property can only be measured in an atom in gaseous state.
X + e− → X−
The electron affinity, Eea, is defined as positive when the resulting ion has a lower energy, i.e. it is an exothermic process that releases energy:
Eea = Einitial − Efinal
Alternatively, electron affinity is often described as the amount of energy required to detach an electron from a singly charged negative ion[1], i.e. the energy change for the process
X− → X + e−
A molecule or atom that has a positive electron affinity is often called an electron acceptor and may undergo charge-transfer reactions.
Contents
Although Eea varies greatly across the periodic table, some patterns emerge. Generally, nonmetals have more positive Eea than metals. Atoms whose anions are more stable than neutral atoms have a greater Eea. Chlorine most strongly attracts extra electrons; mercury most weakly attracts an extra electron. The electron affinities of the noble gases have not been conclusively measured, so they may or may not have slightly negative values.
Eea generally increases across a period (row) in the periodic table. This is caused by the filling of the valence shell of the atom; a group 7A atom releases more energy than a group 1A atom on gaining an electron because it obtains a filled valence shell and therefore is more stable.
A trend of decreasing Eea going down the groups in the periodic table would be expected. The additional electron will be entering an orbital farther away from the nucleus, and thus would experience a lesser effective nuclear charge. However, a clear counterexample to this trend can be found in group 2A, and this trend only applies to group 1A atoms. Electron affinity follows the trend of electronegativity. Fluorine (F) has a higher electron affinity than oxygen and so on.
The Relationship Between Structure and Acidity
Diposting oleh
Vebria Ardina
on Minggu, 27 Maret 2011
Label:
organic chemistry 1
/
Comments: (0)
the strenght of acid and base
Diposting oleh
Vebria Ardina
Label:
organic chemistry 1
/
Comments: (0)
The order of acid strength solution of HCl, HClO, HClO2, HClO3, and HClO4
How you can indulge in acid strength of the compounds of HCl, HClO, HClO2, HClO3, and HClO4? What parameters you can use to measure the degree of acidity of these compounds?
Trend strength above acid can be determined with the following parameters:
Acid strength will be greater with increasing number of oxygen atoms attached to the center.
Acid strength will be even greater with the increasing size of the central atom oxidation number (in kasusini the Cl atom)
By using the above parameters of course you can sort the acid strength of the above compounds is not it? To know "Kenapanya" then you can read the following description and Structure acids are:
Qualitative explanation
According to the Bronsted-Lowry acid is a proton donor, so the acid strength is determined by how easily a species to donate protons. The easier a proton donating species, then the acid will become stronger as well as vice versa. Easy or not an acid species to donate protons can be seen from how much the price of Ka and how much acid is ionized in solution.
We note compounds HClO, HClO2, HClO3, and HClO4 are ionized in water by reaction as follows:
HClO + H2O -> H3O + + clo-
HClO2 + H2O -> H3O + + ClO2-
HClO3 + H2O -> H3O + + ClO3-
HClO4 + H2O -> H3O + + ClO4-
The greater the number of ionized species, the amino acid will be stronger and vice versa. How can we determine the above acids, which one would ionized perfect and which are ionized in part to our use in determining the strength of acid?
How you can use is to determine the stability of residual acid anions in the solution of anion clo-, ClO2-, ClO3-, and ClO4-. The more stable anionnya the more acidic ionized and automatically stronger acidic.
How you can determine the stability of these anions? The answer is by seeing how these anions distribute the negative charge (or in other words look at the structure resonance). The greater the amount of oxygen atoms above the more stable the anion, because the more the number of oxygen atoms that can accept the negative charge distribution, this also means that the anion has a lot of resonance structures.
As an illustration, we carry a lighter load with 4 people instead of carrying the same load with two people. For the above case let's just bebanya is a negative charge, ClO4-ions can distribute the negative charge on the 4 oxygen atoms while the ion-ClO3 can only distribute the negative charge on the 3 oxygen atoms, two to ClO2-ion, and unfortunately ion-CLO can not distribute content negative, so that ClO4-anions is much more stable than the other.
Thus the above sequence is a stable anion-ClO4> ClO3-> ClO2-> clo-. Remember the more stable anion which means more acid is ionized so that the greater acid strength by sebabitu acid sequence of the greatest strengths is HClO4> HClO3> HClO2> HClO.
By looking at the price of Ka / pKa
Price pKa of the acid above is:
HCl pKa = -8
HClO pKa = 7.53
HClO2 pKa = 2
pKa HClO3 = -1
pKa HClO4 = -10
The lower the price pKa the stronger acid, so according to the price above the acid strength of the largest are HClO4> HCl> HClO3> HClO2> HClO. From the data above the price pKa HCl, HClO3, and HClO4 is negative because these acids are strong acids. HCl strength is almost the same can be said with HClO4, this possibility is because HCl in the form of solution [HCl (aq)] as an ionic compound is so easy to let go of its protons HCl. (Gaseous HCl is a weak acid because the H-Cl bond in the form of gas is covalent).
Note:
HCl in the first discussion I was not included because we can not compare the strength of hydrochloric acid in kualittaif with HClO, HClO2, HClO3, and HClO4 is an oxy acid. We are more easily compare the strength of acidity of HCl is qualitatively with HI, HBr, or HF.
posted by : Name : vebria Ardina
NIM : RSA1C110020
class : ISSTE of Chemistry
How you can indulge in acid strength of the compounds of HCl, HClO, HClO2, HClO3, and HClO4? What parameters you can use to measure the degree of acidity of these compounds?
Trend strength above acid can be determined with the following parameters:
Acid strength will be greater with increasing number of oxygen atoms attached to the center.
Acid strength will be even greater with the increasing size of the central atom oxidation number (in kasusini the Cl atom)
By using the above parameters of course you can sort the acid strength of the above compounds is not it? To know "Kenapanya" then you can read the following description and Structure acids are:
Qualitative explanation
According to the Bronsted-Lowry acid is a proton donor, so the acid strength is determined by how easily a species to donate protons. The easier a proton donating species, then the acid will become stronger as well as vice versa. Easy or not an acid species to donate protons can be seen from how much the price of Ka and how much acid is ionized in solution.
We note compounds HClO, HClO2, HClO3, and HClO4 are ionized in water by reaction as follows:
HClO + H2O -> H3O + + clo-
HClO2 + H2O -> H3O + + ClO2-
HClO3 + H2O -> H3O + + ClO3-
HClO4 + H2O -> H3O + + ClO4-
The greater the number of ionized species, the amino acid will be stronger and vice versa. How can we determine the above acids, which one would ionized perfect and which are ionized in part to our use in determining the strength of acid?
How you can use is to determine the stability of residual acid anions in the solution of anion clo-, ClO2-, ClO3-, and ClO4-. The more stable anionnya the more acidic ionized and automatically stronger acidic.
How you can determine the stability of these anions? The answer is by seeing how these anions distribute the negative charge (or in other words look at the structure resonance). The greater the amount of oxygen atoms above the more stable the anion, because the more the number of oxygen atoms that can accept the negative charge distribution, this also means that the anion has a lot of resonance structures.
As an illustration, we carry a lighter load with 4 people instead of carrying the same load with two people. For the above case let's just bebanya is a negative charge, ClO4-ions can distribute the negative charge on the 4 oxygen atoms while the ion-ClO3 can only distribute the negative charge on the 3 oxygen atoms, two to ClO2-ion, and unfortunately ion-CLO can not distribute content negative, so that ClO4-anions is much more stable than the other.
Thus the above sequence is a stable anion-ClO4> ClO3-> ClO2-> clo-. Remember the more stable anion which means more acid is ionized so that the greater acid strength by sebabitu acid sequence of the greatest strengths is HClO4> HClO3> HClO2> HClO.
By looking at the price of Ka / pKa
Price pKa of the acid above is:
HCl pKa = -8
HClO pKa = 7.53
HClO2 pKa = 2
pKa HClO3 = -1
pKa HClO4 = -10
The lower the price pKa the stronger acid, so according to the price above the acid strength of the largest are HClO4> HCl> HClO3> HClO2> HClO. From the data above the price pKa HCl, HClO3, and HClO4 is negative because these acids are strong acids. HCl strength is almost the same can be said with HClO4, this possibility is because HCl in the form of solution [HCl (aq)] as an ionic compound is so easy to let go of its protons HCl. (Gaseous HCl is a weak acid because the H-Cl bond in the form of gas is covalent).
Note:
HCl in the first discussion I was not included because we can not compare the strength of hydrochloric acid in kualittaif with HClO, HClO2, HClO3, and HClO4 is an oxy acid. We are more easily compare the strength of acidity of HCl is qualitatively with HI, HBr, or HF.
posted by : Name : vebria Ardina
NIM : RSA1C110020
class : ISSTE of Chemistry
rasa asin pada air laut
Diposting oleh
Vebria Ardina
Label:
chemistry is our life
/
Comments: (0)
Bagaimanakah rasa air laut?? ASIN,, Kenapa rasa air laut asin?? Darimanakah rasa asin itu??
Merasa bingung khan?? Mari kita bicarakan bersama....
Laut adalah kumpulan air asin yang luas dan berhubungan dengan samudra.
Penyebab rasa asin itu adalah garam-garaman yang terkandung di dalam air laut. Air di laut merupakan campuran dari 96,5% air murni dan 3,5% material lainnya seperti garam-garaman, gas-gas terlarut, bahan-bahan organik dan partikel-partikel tak terlarut. Sifat-sifat fisis utama air laut ditentukan oleh 96,5% air murni.Jumlah dari seluruh garam-garaman dalam gram pada setiap kilogram air laut disebut salinitas. Garam adalah senyawa kimia yang terdiri dari dua bagian yang bermuatan positif dan bermuatan negatif yang keduanya saling tarik menarik sehingga membentuk “garam” (bagian yang bermuatan positif dan negatif ini disebut ION). Unsur Garam-garaman utama yang terdapat dalam air laut adalah klorida (55%), natrium (31%), sulfat (8%), magnesium (4%), kalsium (1%), potasium (1%) dan sisanya (kurang dari 1%) teridiri dari bikarbonat, bromida, asam borak, strontium dan florida.
Darimanakah asalnya garam-garamam tersebut??
Sumber utama garam-garaman di laut adalah pelapukan batuan di darat, gas-gas vulkanik dan sirkulasi lubang-lubang hidrotermal (hydrothermal vents) di laut dalam. Bagaimana prosesnya? Kita tahu bahwa laut adalah tempat berkumpulnya semua air; air dari rumah, selokan, sungai, semua air, termasuk air hujan. Kesemuanya mengalir menuju laut, sambil “mencuci” batuan, tanah, semua benda yang dilewatinya, sambil membawa juga ion-ion tadi (semua terjadi dalam proses hidrologis). Belum lagi peristiwa alam seperti letusan gunung api baik yang di daratan maupun di lautan, semua memberi kandungan khlor yang berlimpah bagi lautan. Proses ini terjadi secara terus menerus selama milyaran tahun.
Laut, menurut sejarahnya, terbentuk 4,4 milyar tahun yang lalu, dimana awalnya bersifat sangat asam dengan air yang mendidih (dengan suhu sekitar 100°C) karena panasnya Bumi pada saat itu. Asamnya air laut terjadi karena saat itu atmosfir Bumi dipenuhi oleh Karbondioksida. Keasaman air inilah yang menyebabkan tingginya pelapukan yang terjadi yang menghasilkan garam-garaman yang menyebabkan air laut menjadi asin seperti sekarang ini.
Merasa bingung khan?? Mari kita bicarakan bersama....
Laut adalah kumpulan air asin yang luas dan berhubungan dengan samudra.
Penyebab rasa asin itu adalah garam-garaman yang terkandung di dalam air laut. Air di laut merupakan campuran dari 96,5% air murni dan 3,5% material lainnya seperti garam-garaman, gas-gas terlarut, bahan-bahan organik dan partikel-partikel tak terlarut. Sifat-sifat fisis utama air laut ditentukan oleh 96,5% air murni.Jumlah dari seluruh garam-garaman dalam gram pada setiap kilogram air laut disebut salinitas. Garam adalah senyawa kimia yang terdiri dari dua bagian yang bermuatan positif dan bermuatan negatif yang keduanya saling tarik menarik sehingga membentuk “garam” (bagian yang bermuatan positif dan negatif ini disebut ION). Unsur Garam-garaman utama yang terdapat dalam air laut adalah klorida (55%), natrium (31%), sulfat (8%), magnesium (4%), kalsium (1%), potasium (1%) dan sisanya (kurang dari 1%) teridiri dari bikarbonat, bromida, asam borak, strontium dan florida.
Darimanakah asalnya garam-garamam tersebut??
Sumber utama garam-garaman di laut adalah pelapukan batuan di darat, gas-gas vulkanik dan sirkulasi lubang-lubang hidrotermal (hydrothermal vents) di laut dalam. Bagaimana prosesnya? Kita tahu bahwa laut adalah tempat berkumpulnya semua air; air dari rumah, selokan, sungai, semua air, termasuk air hujan. Kesemuanya mengalir menuju laut, sambil “mencuci” batuan, tanah, semua benda yang dilewatinya, sambil membawa juga ion-ion tadi (semua terjadi dalam proses hidrologis). Belum lagi peristiwa alam seperti letusan gunung api baik yang di daratan maupun di lautan, semua memberi kandungan khlor yang berlimpah bagi lautan. Proses ini terjadi secara terus menerus selama milyaran tahun.
Laut, menurut sejarahnya, terbentuk 4,4 milyar tahun yang lalu, dimana awalnya bersifat sangat asam dengan air yang mendidih (dengan suhu sekitar 100°C) karena panasnya Bumi pada saat itu. Asamnya air laut terjadi karena saat itu atmosfir Bumi dipenuhi oleh Karbondioksida. Keasaman air inilah yang menyebabkan tingginya pelapukan yang terjadi yang menghasilkan garam-garaman yang menyebabkan air laut menjadi asin seperti sekarang ini.
why is the water droplets round?
Diposting oleh
Vebria Ardina
Label:
chemistry is our life
/
Comments: (0)
Why is the water droplets round?
Perhaps these questions are rarely ignored by many. here I will try to uncover this mystery. every drop of water falling to the ground or down it must be round. Spherical shape is the shape and tersimpel populous than all existing forms, such as rectangular or triangular. This simple form makes the water droplets become unstable, and therefore all forms of the elements or anything else always has a tendency to form something more stable. Then the wisdom what is hidden behind this phenomenon. as we know that our earth is round, everything is round always familiar to many people, just for example football, basketball, dsb.Kehidupan it's like a round ball and is always spinning, there is sometimes above and sometimes under . This situation caused the man to do ever mnyombongkan yourself for what has he got, because God was the one who has the truth. This life is also always lead to stability and equilibrium. Everything in this universe will be in equilibrium, namely death. therefore we sembagai a servant who is weak, it should always be ready with his arrival, and always our position to form a more stable from day to day. as shown by the noble gas elements that have remarkable stability. If the element is stable by imitating the number of electrons that are owned by the noble gases, why we do not do it. Indeed the rules created are not made to narrow down / restrain us, but to help us later.
Perhaps these questions are rarely ignored by many. here I will try to uncover this mystery. every drop of water falling to the ground or down it must be round. Spherical shape is the shape and tersimpel populous than all existing forms, such as rectangular or triangular. This simple form makes the water droplets become unstable, and therefore all forms of the elements or anything else always has a tendency to form something more stable. Then the wisdom what is hidden behind this phenomenon. as we know that our earth is round, everything is round always familiar to many people, just for example football, basketball, dsb.Kehidupan it's like a round ball and is always spinning, there is sometimes above and sometimes under . This situation caused the man to do ever mnyombongkan yourself for what has he got, because God was the one who has the truth. This life is also always lead to stability and equilibrium. Everything in this universe will be in equilibrium, namely death. therefore we sembagai a servant who is weak, it should always be ready with his arrival, and always our position to form a more stable from day to day. as shown by the noble gas elements that have remarkable stability. If the element is stable by imitating the number of electrons that are owned by the noble gases, why we do not do it. Indeed the rules created are not made to narrow down / restrain us, but to help us later.
An introduction to organic reaction
Diposting oleh
Vebria Ardina
on Minggu, 20 Maret 2011
Label:
organic chemistry 1
/
Comments: (0)
In this lessonWe learn about an introduction to organic reaction : Acid and Bases, and I less understood about
Question : What is relationship between structur and acidity?
According My opinion The strength of an acid depends on the extent to which a proton can be separated from it and transferred to a base.Breaking a bond to the proton Þ the strength of the bond to the proton is the dominating effect.
The Acidity of Carboxylic Acids
Carboxylic acids are much more acidic than the corresponding alcohols: pKas for R–COOH are in the range of 3-5;.pKas for R–OH are in the range of 15-18.
An Explanation Based on Resonance Effects
Resonance stabilized acetate anion: The greater stabilization of the carboxylate anion (relative to the acid) lowers the free energy of the anion and thereby decreases the positive free-energy change required for the ionization. Any factor that makes the free-energy change for the ionization of an acid less positive (or more negative) makes the acid stronger.
An Explanation Based on Inductive Effects
The inductive effect of the carbonyl group (C=O group) is responsible for the acidity of carboxylic acids.
Organic Compounds as Bases
An organic compound contains an atom with an unshared electron pair is a potential base. The p bond of an alkene can act as a base.
The Acidity of Carboxylic Acids
Carboxylic acids are much more acidic than the corresponding alcohols: pKas for R–COOH are in the range of 3-5;.pKas for R–OH are in the range of 15-18.
An Explanation Based on Resonance Effects
Resonance stabilized acetate anion: The greater stabilization of the carboxylate anion (relative to the acid) lowers the free energy of the anion and thereby decreases the positive free-energy change required for the ionization. Any factor that makes the free-energy change for the ionization of an acid less positive (or more negative) makes the acid stronger.
An Explanation Based on Inductive Effects
The inductive effect of the carbonyl group (C=O group) is responsible for the acidity of carboxylic acids.
Organic Compounds as Bases
An organic compound contains an atom with an unshared electron pair is a potential base. The p bond of an alkene can act as a base.
Posted by : NAME : VEBRIA ARDINA
NIM : RSA1CI10020
ANGKATAN 2010
CLASS : ISSTE of CHEMISTRY
Alkene
Diposting oleh
Vebria Ardina
on Sabtu, 19 Maret 2011
Label:
organic chemistry 1
/
Comments: (0)
Why are alkenes are relatively stable compounds, but are more reactive than alkanes?
Ethylene (ethene), showing the pi bond in green. According my opinion it is because Alkenes are relatively stable compounds, but are more reactive than alkanes due to the presence of a carbon-carbon pi-bond. It is also attributed to the presence of pi-electrons in the molecule. The majority of the reactions of alkenes involve the rupture of this pi bond, forming new single bonds.
Like single covalent bonds, double bonds can be described in terms of overlapping atomic orbitals, except that, unlike a single bond (which consists of a single sigma bond), a carbon-carbon double bond consists of one sigma bond and one pi bond. This double bond is stronger than a single covalent bond (611 kJ/mol for C=C vs. 347 kJ/mol for C—C)[1] and also shorter with an average bond length of 1.33 Angstroms (133 pm).
Each carbon of the double bond uses its three sp² hybrid orbitals to form sigma bonds to three atoms. The unhybridized 2p atomic orbitals, which lie perpendicular to the plane created by the axes of the three sp² hybrid orbitals, combine to form the pi bond. This bond lies outside the main C—C axis, with half of the bond on one side and half on the other.
Rotation about the carbon-carbon double bond is restricted because it involves breaking the pi bond, which requires a large amount of energy (264 kJ/mol in ethylene). As a consequence, substituted alkenes may exist as one of two isomers, called cis or trans isomers. More complex alkenes may be named using the E-Z notation, used to describe molecules having three or four different substituents (side groups). For example, of the isomers of butene, the two methyl groups of (Z)-but-2-ene (aka cis-2-butene) face the same side of the double bond, and in (E)-but2-ene (aka trans-2-butene) the methyl groups face the opposite side. These two isomers of butene are slightly different in their chemical and physical properties.
It is certainly not impossible to twist a double bond. In fact, a 90° twist requires an energy approximately equal to half the strength of a pi bond. The misalignment of the p orbitals is less than expected because pyramidalization takes place (See: pyramidal alkene). trans-Cyclooctene is a stable strained alkene and the orbital misalignment is only 19° with a dihedral angle of 137° (normal 120°) and a degree of pyramidalization of 18°. This explains the dipole moment of 0.8 D for this compound (cis-isomer 0.4 D) where a value of zero is expected.[3] The trans isomer of cycloheptene is only stable at low temperatures.
As predicted by the VSEPR model of electron pair repulsion, the molecular geometry of alkenes includes bond angles about each carbon in a double bond of about 120°. The angle may vary because of steric strain introduced by nonbonded interactions created by functional groups attached to the carbons of the double bond. For example, the C-C-C bond angle in propylene is 123.9°.
The physical properties of alkenes are comparable with those of alkanes. The physical state depends on molecular mass (gases from ethene to butene - liquids from pentene onwards). The simplest alkenes, ethene, propene and butene are gases. Linear alkenes of approximately five to sixteen carbons are liquids, and higher alkenes are waxy solids.
Alkenes react in many addition reactions, which occur by opening up the double-bond. Most addition reactions to alkenes follow the mechanism of electrophilic addition. Examples of addition reactions are hydrohalogenation, halogenation, halohydrin formation, oxymercuration, hydroboration, dichlorocarbene addition, Simmons-Smith reaction, catalytic hydrogenation, epoxidation, radical polymerization and hydroxylation.Like single covalent bonds, double bonds can be described in terms of overlapping atomic orbitals, except that, unlike a single bond (which consists of a single sigma bond), a carbon-carbon double bond consists of one sigma bond and one pi bond. This double bond is stronger than a single covalent bond (611 kJ/mol for C=C vs. 347 kJ/mol for C—C)[1] and also shorter with an average bond length of 1.33 Angstroms (133 pm).
Each carbon of the double bond uses its three sp² hybrid orbitals to form sigma bonds to three atoms. The unhybridized 2p atomic orbitals, which lie perpendicular to the plane created by the axes of the three sp² hybrid orbitals, combine to form the pi bond. This bond lies outside the main C—C axis, with half of the bond on one side and half on the other.
Rotation about the carbon-carbon double bond is restricted because it involves breaking the pi bond, which requires a large amount of energy (264 kJ/mol in ethylene). As a consequence, substituted alkenes may exist as one of two isomers, called cis or trans isomers. More complex alkenes may be named using the E-Z notation, used to describe molecules having three or four different substituents (side groups). For example, of the isomers of butene, the two methyl groups of (Z)-but-2-ene (aka cis-2-butene) face the same side of the double bond, and in (E)-but2-ene (aka trans-2-butene) the methyl groups face the opposite side. These two isomers of butene are slightly different in their chemical and physical properties.
It is certainly not impossible to twist a double bond. In fact, a 90° twist requires an energy approximately equal to half the strength of a pi bond. The misalignment of the p orbitals is less than expected because pyramidalization takes place (See: pyramidal alkene). trans-Cyclooctene is a stable strained alkene and the orbital misalignment is only 19° with a dihedral angle of 137° (normal 120°) and a degree of pyramidalization of 18°. This explains the dipole moment of 0.8 D for this compound (cis-isomer 0.4 D) where a value of zero is expected.[3] The trans isomer of cycloheptene is only stable at low temperatures.
As predicted by the VSEPR model of electron pair repulsion, the molecular geometry of alkenes includes bond angles about each carbon in a double bond of about 120°. The angle may vary because of steric strain introduced by nonbonded interactions created by functional groups attached to the carbons of the double bond. For example, the C-C-C bond angle in propylene is 123.9°.
The physical properties of alkenes are comparable with those of alkanes. The physical state depends on molecular mass (gases from ethene to butene - liquids from pentene onwards). The simplest alkenes, ethene, propene and butene are gases. Linear alkenes of approximately five to sixteen carbons are liquids, and higher alkenes are waxy solids.
Posted by : NAME : VEBRIA ARDINA
NIM : RSA1C110020
STUDY PROGRAMME : ISSTE OF CHEMISTRY
URL BLOG : http://vebriasanjaya.blogspot.com
Reaction of alkane
Diposting oleh
Vebria Ardina
on Jumat, 18 Maret 2011
Label:
organic chemistry 1
/
Comments: (0)
Reaction of alkene
1. Why are mostly alkenes compound are inert?
Answer : Because they bonding are saturated (all their rooms is filled by electron).
2. How to break down the bonding of 3, 4, 5 trimethyl nonane into n- butene?
Answer : The first we must find the knots of reactions or centre of reactions agree with the result which we want, like normal butene about less or more than 60 %
H H H H H H H H H
H C C C C C C C C C H
H H CH3 CH3 CH3 H H H H
H H H H H H H H H
H C C C C C C C C C H
H H CH3 CH3 CH3 H H H H
From the node that has the mark on so we can get some stricture of n-butene, namely
H H H
H C C C H
H H CH3
n- butena
H H
H C C H
CH3 CH3
n- butena
H H H H
H C C C C H
H H H H
n- butena
H C C H
CH3 CH3
n- butena
H H H H
H C C C C H
H H H H
n- butena
:
Isomerism
Diposting oleh
Vebria Ardina
on Minggu, 13 Maret 2011
Label:
organic chemistry 1
/
Comments: (0)
Compounds that have same molecular formula but different structures are called Isomers. There are many kinds of isomers in organic compounds,
Constitutional Isomers
In the form of n-butane and isobutane, constitutional isomers have the same molecular formula, but different connectivities. The difference between the two butanes lies in which atoms are connected to which (the connectivities), and this is best seen by examining the carbon skeletons.
n-butane
isobutane
Note that n- butane is astright chain, whereas isobutane has one branch. The two isomers are different compounds and have different physical and chemical properties, including quite different molecular shapes. A mixture of the two isomers can be physically separated into two pure components. the only common feature they share is their molecular formula C4H10.
Posted By :
Name : Vebria Ardina
NIM : RSA1C110020
Class : Chemistry Of PGSBI
angkatan : 2010
Nama blog : http://vebriasanjaya.blogspot.com/reguler2009@gmail.com
Constitutional Isomers
In the form of n-butane and isobutane, constitutional isomers have the same molecular formula, but different connectivities. The difference between the two butanes lies in which atoms are connected to which (the connectivities), and this is best seen by examining the carbon skeletons.
n-butane
isobutane
Note that n- butane is astright chain, whereas isobutane has one branch. The two isomers are different compounds and have different physical and chemical properties, including quite different molecular shapes. A mixture of the two isomers can be physically separated into two pure components. the only common feature they share is their molecular formula C4H10.
Posted By :
Name : Vebria Ardina
NIM : RSA1C110020
Class : Chemistry Of PGSBI
angkatan : 2010
Nama blog : http://vebriasanjaya.blogspot.com/reguler2009@gmail.com
Alkanes
Diposting oleh
Vebria Ardina
on Kamis, 10 Maret 2011
Label:
organic chemistry 1
/
Comments: (0)
Why are alkanes inert?
Alkanes (also known as paraffins or saturated hydrocarbons) are chemical compounds that consist only of the elements carbon (C) and hydrogen (H) (i.e., hydrocarbons), wherein these atoms are linked together exclusively by single bonds (i.e., they are saturated compounds). Alkanes belong to a homologous series of organic compounds in which the members differ by a constant relative molecular mass of 14.
Each carbon atom must have 4 bonds (either C-H or C-C bonds), and each hydrogen atom must be joined to a carbon atom (H-C bonds). A series of linked carbon atoms is known as the carbon skeleton or carbon backbone. In general, the number of carbon atoms is often used to define the size of the alkane (e.g., C2-alkane).
An Alkyl group, generally abbreviated with the symbol R, is a functional group or side-chain that, like an alkane, consists solely of single-bonded carbon and hydrogen atoms, for example a methyl or ethyl group.
In general, alkanes show a relatively low reactivity, because their C bonds are relatively stable and cannot be easily broken. Unlike most other organic compounds, they possess no functional groups.
They react only very poorly with ionic or other polar substances. The acid dissociation constant (pKa) values of all alkanes are above 60, hence they are practically inert to acids and bases (see: carbon acids). This inertness is the source of the term paraffins (with the meaning here of "lacking affinity"). In crude oil the alkane molecules have remained chemically unchanged for millions of years.
However redox reactions of alkanes, in particular with oxygen and the halogens, are possible as the carbon atoms are in a strongly reduced condition; in the case of methane, the lowest possible oxidation state for carbon (−4) is reached. Reaction with oxygen (if an amount of the least is enough to meet the reaction stoichometry) leads to combustion without any smoke; with halogens, substitution. In addition, alkanes have been shown to interact with, and bind to, certain transition metal complexes in (See: carbon-hydrogen bond activation).
Free radicals, molecules with unpaired electrons, play a large role in most reactions of alkanes, such as cracking and reformation where long-chain alkanes are converted into shorter-chain alkanes and straight-chain alkanes into branched-chain isomers.
In highly branched alkanes, the bond angle may differ significantly from the optimal value (109.5°) in order to allow the different groups sufficient space. This causes a tension in the molecule, known as steric hindrance, and can substantially increase the reactivity.
The simplest possible alkane (the parent molecule) is methane, CH4. There is no limit to the number of carbon atoms that can be linked together, the only limitation being that the molecule is acyclic, is saturated, and is a hydrocarbon. Saturated oils and waxes are examples of larger alkanes where the number of carbons in the carbon backbone tends to be greater than 10.
Alkanes are not very reactive and have little biological activity. Alkanes can be viewed as a molecular tree upon which can be hung the interesting biologically active/reactive portions (functional groups) of the molecule.
Posted by :
Name : VEBRIA ARDINA
NIM : RSA1C110020
ANGKATAN : 2010
KELAS : PPG-MIPA BI
NAMA BLOG : http://vebriasanjaya.blogspot.com/reguler2009@gmail.com
vebriardina@yahoo.co.id
kenonpolaran Hidrocarbon
Diposting oleh
Vebria Ardina
on Minggu, 06 Maret 2011
Label:
organic chemistry 1
/
Comments: (0)
reguler2009@gmail.comvebriaardina@yahooo.co.idwhichever is lowest kenonpolarannya among alkanes, alkenes, and alkyne?
Among the three alkanes, alkenes, and alkyne lowest alkyne nonpolarnya is because among the three compounds on the most easily soluble in water is alkyne and alkenes and alkanes are compounds that can not dissolve in water.
Posted by:
Name : VEBRIA ARDINA
NIM : RSA1C110020
Posted by:
Name : VEBRIA ARDINA
NIM : RSA1C110020
Ethylene gas
Diposting oleh
Vebria Ardina
Label:
organic chemistry 1
/
Comments: (0)
Ethylene gas (C2H4) is an odorless, colorless gas that exists in nature and is also created by man-made sources. Not easily detectable, it exists where produce is stored. In nature, the largest producers are plant and plant products (ie. fruits, vegetables and floral products) which produce ethylene within their tissues and release it into the surrounding atmosphere. It is also a by-product of man-made processes, such as combustion.
Ethylene, also known as the 'death' or 'ripening hormone' plays a regulatory role in many processes of plant growth, development and eventually death. Fruits, vegetables and flowers contain receptors which serve as bonding sites to absorb free atmospheric ethylene molecules. The common practice of placing a tomato, avocado or banana in a paper bag to hasten ripening is an example of the action of ethylene on produce. Increased levels of ethylene contained within the bag, released by the produce itself, serves as a stimulant after reabsorption to initiate the production of more ethylene. The overall effect is to hasten ripening, aging and eventually spoilage. A refrigerator acts in much the same way. Kept closed to retain the desired temperature, it also enables an increased concentration of ethylene to accumulate. Any closed environment, such as a truck trailer, shipping container or warehouse, will have a similar effect.
Ethylene is a plant hormone that differs from other plant hormones in being a gas. It has the molecular structure: H2C=CH2 When fruits approach maturity, they release ethylene. Ethylene promotes the ripening of fruit. Among the many changes that ethylene causes is the destruction of chlorophyll. With the breakdown of chlorophyll, the red and/or yellow pigments in the cells of the fruit are unmasked and the fruit assumes its ripened color. The presence of ethylene is probably detected by transmembrane receptors (protein that passes one or more times through the lipid bilayer of a cell membrane) in the surface of the plasma membrane of the cells.
EFFECTS
Fruit Ripening
Abscission; leaf flower fruits (thinning, harvesting)
Epinasty
Triple Resonses
Hook Closure Maintenance
Initiates Germination in Grains
Activates dormant buds (potatoes in storage)
Stem elongation in deep-water rice
Induces Flowering in Pineapple
Promotes Female Expression in Flowers
Flower and Leaf Senescence: Ag preventative (vase life)
While ethylene is invaluable due to its ability to initiate the ripening process in several fruits, it can also be very harmful to many fruits, vegetables, flowers, and plants by accelerating the aging process and decreasing the product quality and shelf life. The degree of damage depends upon the concentration of ethylene, length of exposure time, and product temperature. One of the following methods should be used to ensure that ethylene-sensitive produce is not exposed: a) Ethylene producing items (such as apples, avocados, bananas, melons, peaches, pears, and tomatoes) should be stored separately from ethylene-sensitive ones (broccoli, cabbage, cauliflower, leafy greens, lettuce, etc.). Also, ethylene is emitted by engines. Propane, diesel, and gasoline powered engines all produce ethylene in amounts large enough to cause damage to the ethylene-sensitive produce items mentioned; b) Ventilate the storage area, preferably to the outside of the warehouse, on a continuous or regular basis to purge the air of any ethylene; c) Remove ethylene with ethylene absorbing filters. These have been proven in reducing and maintaining low ethylene levels. If ethylene damage is suspected, a quick and easy way to detect ethylene levels is with hand held sensor tubes. This will indicate if the above steps should be followed.
ethylene gas is effective at 0.1 to 1 ppm. One part of ethylene per million parts of air that's one cupful of ethylene gas in 62,000 gallons of air - is enough to promote the ripening process in fruits.
why the fruit is ripe containing ethylene gas??if the fruit is not ripe d mixed with fruit that is ripe, it is not yet ripe fruit will cook faster. why did that happen?
Listen
Read phonetically
Dictionary - View detailed dictionary
ethylene gas is able to solve the chlorophyll in the young fruit until the fruit has only resulted in xantofil and xarotein or substance that makes skin becomes red or orange fruit. because chlorophyll has been reduced by ethylene gas ....
only that the function of ethylene in ripening fruit, if problems flavors, from sour to sweet when the fruit is ripe, it depends on whether or not split sel2 perfect cork on the fruit or it could be caused by genetic factors.stimulated fruit ripening by ethylene gas that diffuses into the intercellular spaces of the fruit. Gas can also diffuse through the air from one fruit to another fruit, fruit will mature more quickly if the fruit is stored in a plastic bag which resulted in accumulated ethylene gas.
posted by
name : VEBRIA ARDINA
NIM : RSA1C110020
Angkatan : 2010
Kelas : PPG-MIPA-BI 2010 (CHEMISTRY)
Nama Blog : http://vebriasanjaya.blogspot.com/
Listen
Read phonetically
Dictionary - View detailed dictionary
Listen
Read phonetically
Dictionary - View detailed dictionary
Diposting oleh
Vebria Ardina
on Sabtu, 05 Maret 2011
Label:
Hydrocarbon is non polar
/
Comments: (0)
Hydrocarbons
| Hydrocarbons are the simplest organic compounds . Containing only carbon and hydrogen, they can be straight-chain, branched chain, or cyclic molecules. Carbon tends to form four bonds in a tetrahedral geometry. Hydrocarbon derivatives are formed when there is a substitution of a functional group at one or more of these positions. | 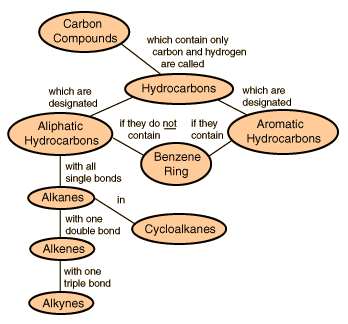 |
POLAR AND NONPOLAR COMPOUNDS
Depending on the relative electronegativities of the two atoms sharing electrons, there may be partial transfer of electron density from one atom to the other. When the electronegativities are not equal, electrons are not shared equally and partial ionic charges develop. The greater the electronegativity difference, the more ionic the bond is. Bonds that are partly ionic are called polar covalent bonds.
Nonpolar covalent bonds, with equal sharing of the bond electrons, arise when the electronegativities of the two atoms are equal.
Nonpolar covalent bonds, with equal sharing of the bond electrons, arise when the electronegativities of the two atoms are equal.
 |
- A bond between 2 nonmetal atoms that have the same electronegativity and therefore have equal sharing of the bonding electron pair
- Example: In H-H each H atom has an electronegativity value of 2.1, therefore the covalent bond between them is considered nonpolar
Polar Covalent Bond
- A bond between 2 nonmetal atoms that have different electronegativities and therefore have unequal sharing of the bonding electron pair
- Example: In H-Cl, the electronegativity of the Cl atom is 3.0, while that of the H atom is 2.1
- The result is a bond where the electron pair is displaced toward the more electronegative atom. This atom then obtains a partial-negative charge while the less electronegative atom has a partial-positive charge.This separation of charge or bond dipole can be illustrated using an arrow with the arrowhead directed toward the more electronegative atom.

The Greek letter delta indicates "partially".
- Within a molecule each polar bond has a bond dipole
- A polar molecule always contains polar bonds, but some molecules with polar bonds are nonpolar.
- A molecule in which the bond dipoles present do not cancel each other out and thus results in a molecular dipole.(see below). Cancellation depends on the shape of the molecule or Stereochemistry and the orientation of the polar bonds.
Molecular Dipole
- A result of the bond dipoles in a molecule.
- Bond dipoles may or may not cancel out thereby producing either molecules that are nonpolar, if they cancel, or polar, if they do not cancel
- Examples:
- CO2 is a linear molecule with 2 bond dipoles that are equal and oppositely directed therefore the bond polarities cancel and the molecule is nonpolar.
- HCN is a linear molecule with 2 bond dipoles that are in the same direction and are not equal therefore the bond polarities do not cancel and the molecule is polar
- More examples can be found on the Table: Stereochemistry of Some Common Molecules
Why are hydrocarbon nonpolar?
Hydrocarbon molecules are made of a carbon backbone with hydrogen attachments. Based on electronegativity, neither of these elements create polarity (electrons being pulled towards one pole). When you have a large difference in electronegativity (O-H) then you create polarity. Carbon and Hydrogen are too close in their electronegativity and therefore share electrons equally.