Hydrocarbons
| Hydrocarbons are the simplest organic compounds . Containing only carbon and hydrogen, they can be straight-chain, branched chain, or cyclic molecules. Carbon tends to form four bonds in a tetrahedral geometry. Hydrocarbon derivatives are formed when there is a substitution of a functional group at one or more of these positions. | 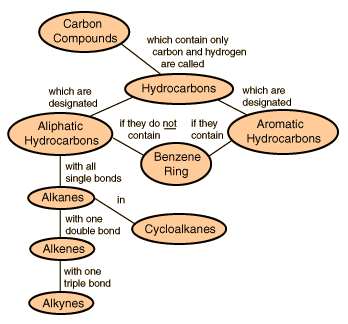 |
POLAR AND NONPOLAR COMPOUNDS
Depending on the relative electronegativities of the two atoms sharing electrons, there may be partial transfer of electron density from one atom to the other. When the electronegativities are not equal, electrons are not shared equally and partial ionic charges develop. The greater the electronegativity difference, the more ionic the bond is. Bonds that are partly ionic are called polar covalent bonds.
Nonpolar covalent bonds, with equal sharing of the bond electrons, arise when the electronegativities of the two atoms are equal.
Nonpolar covalent bonds, with equal sharing of the bond electrons, arise when the electronegativities of the two atoms are equal.
 |
- A bond between 2 nonmetal atoms that have the same electronegativity and therefore have equal sharing of the bonding electron pair
- Example: In H-H each H atom has an electronegativity value of 2.1, therefore the covalent bond between them is considered nonpolar
Polar Covalent Bond
- A bond between 2 nonmetal atoms that have different electronegativities and therefore have unequal sharing of the bonding electron pair
- Example: In H-Cl, the electronegativity of the Cl atom is 3.0, while that of the H atom is 2.1
- The result is a bond where the electron pair is displaced toward the more electronegative atom. This atom then obtains a partial-negative charge while the less electronegative atom has a partial-positive charge.This separation of charge or bond dipole can be illustrated using an arrow with the arrowhead directed toward the more electronegative atom.

The Greek letter delta indicates "partially".
- Within a molecule each polar bond has a bond dipole
- A polar molecule always contains polar bonds, but some molecules with polar bonds are nonpolar.
- A molecule in which the bond dipoles present do not cancel each other out and thus results in a molecular dipole.(see below). Cancellation depends on the shape of the molecule or Stereochemistry and the orientation of the polar bonds.
Molecular Dipole
- A result of the bond dipoles in a molecule.
- Bond dipoles may or may not cancel out thereby producing either molecules that are nonpolar, if they cancel, or polar, if they do not cancel
- Examples:
- CO2 is a linear molecule with 2 bond dipoles that are equal and oppositely directed therefore the bond polarities cancel and the molecule is nonpolar.
- HCN is a linear molecule with 2 bond dipoles that are in the same direction and are not equal therefore the bond polarities do not cancel and the molecule is polar
- More examples can be found on the Table: Stereochemistry of Some Common Molecules
Why are hydrocarbon nonpolar?
Hydrocarbon molecules are made of a carbon backbone with hydrogen attachments. Based on electronegativity, neither of these elements create polarity (electrons being pulled towards one pole). When you have a large difference in electronegativity (O-H) then you create polarity. Carbon and Hydrogen are too close in their electronegativity and therefore share electrons equally.


0 komentar:
Posting Komentar